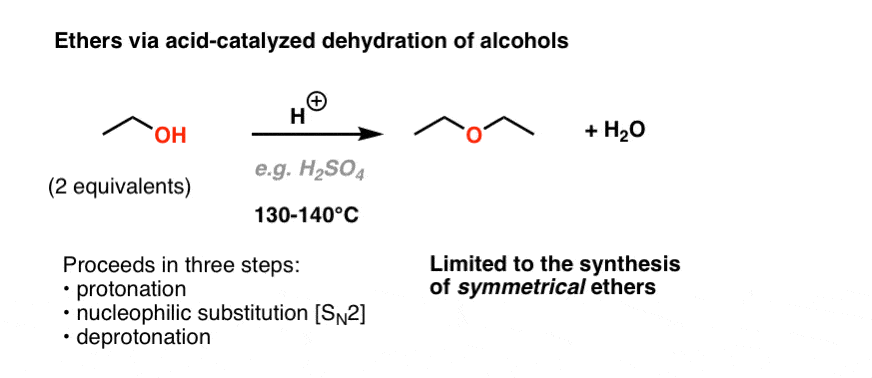
Table of Content
The challenges that contemporary marketers encounter are examined in this market research study on High Purity Sulfuric Acid. The primary problem is the increased level of market rivalry. This study offers solutions to these issues in addition to a thorough analysis of the industry. Watch the full video below to learn the electrochemical method. You'll need some carbon electrodes , copper wire, tape, power source, copper sulfate and filter.
For the purposes of this experiment, it will be easiest to use either sodium nitrate, potassium nitrate, or ammonium nitrate. All of these chemicals are readily available at any online chemistry supply store. Use a pH strip to test the juice - it should be at about 2 or 3 on the pH scale.
Make iodine from hydrochloric acid and H2O2
Alkaline batteries leak a chemical that registers as a base on the pH scale, rather than acid. As a result, a battery leak should be cleaned using a moderate household acid such as vinegar or lemon juice. Using distilled water to replenish your batteries is one of the most straightforward options. A flooded deep cycle battery’s electrolyte is a mixture of acid and water. It is recommended to use distilled or deionized water since it is devoid of extra minerals found in conventional tap water. Sulfuric acid is the sole electrolyte that may be used in a lead-acid battery.
Look up “Blue Moonshine” on YouTube, “How to make concentrated sulfuric acid at home”. This is a very easy DIY that involves cheap household products, and you can end up with 98% sulfuric acid. It’s crucial to remember that sulfuric acid should never be added to a battery. Batteries simply consume water during regular operation, not sulfuric acid.
Make elemental sulfur (sulphur)
For a few seconds, the baking soda will react by bubbling up violently. The acidic corrosion on the battery terminals will be neutralized by the interaction between the baking soda and water combination and the acid, making it safe to handle. The vinegar will react with the chemical components within the battery, causing serious damage.

Dr. Helmenstine holds a Ph.D. in biomedical sciences and is a science writer, educator, and consultant. Intraday Data provided by FACTSET and subject to terms of use. Historical and current end-of-day data provided by FACTSET. Real-time last sale data for U.S. stock quotes reflect trades reported through Nasdaq only.
Petroleum industry
If needed, blame the smell on the nearest oil refinery. We make concentrated sulfuric acid from sodium metabisulfite, hydrochloric acid and an oxidant such as hydrogen peroxide or nitric acid. She has taught science courses at the high school, college, and graduate levels. Turn on your burner or hot plate, then sit back and watch the magic happen. As the acid mixture heats up, the nitrate salt and sulfuric acid will react to produce bisulfate and liquid nitric acid. The nitric acid will then be vaporized and move into the condenser, where it will cool into small droplets that will drain little-by-little into the empty container.

But "vitriol" is not just meant as cruel and bitter criticism. The word is a homonym and has an even older meaning — sulfuric acid. An important usage of sulphuric acid is in potato harvesting. The manufacture of rayon fibers requires the use of sulphuric acid. To make a slurry with various hues, sulphuric acid is fed to dyeing machines.
For homebrew chemists, it might have been a little difficult to build. This means that sulfuric acid will dissolve in water and can be diluted to different strengths depending upon the intended application. Sulfuric acid is a colorless, oily liquid that is water soluble when heated. Dilute sulphuric acid is an odorless, colorless mineral acid. It is used as a hydrating agent, unlike concentrated sulphuric acid.
Sulphuric acid serves as an electrode in electrochemical reactions. Sulphuric acid is employed in producing a number of diverse esters and diesters, including dimethyl sulphate, a particularly potent alkylating agent. Seek immediate medical attention if you notice any burning, swelling, itching, or irritation as a result of exposure to chemicals. If left untreated, chemical burns can lead to more serious symptoms, including shock.
If the coffee filter paper gets clogged, empty the liquid into a beaker, replace the coffee filter, and continue to pour the solution through the filter. It may take several attempts to fully filter the liquid. Evaporation causes water to evaporate from the electrolyte, which is a combination of around one part sulfuric acid and two parts water in older battery designs. If you have one, check it twice a year and only add distilled water as necessary. Sulphuric acid is a powerful mineral acid that at all concentrations is soluble in water. Sulphuric acid contains two atoms of hydrogen, one atom of sulphur, and four atoms of oxygen.
Heat this solution on medium heat to evaporate the water from the beaker. Stir the solution regularly while it is heating up, but don't allow it to boil. As the volume of the solution goes down, you will see it start to turn opaque. Wait until the volume goes down to about 70 millilitres (0.30 c), then take it off the heat. Add an eyedropper filled with 10% strength sodium hydroxide and test it again. The sodium hydroxide will neutralize the acidity of the lemon juice.
The essential components of the battery chemistry are sulfuric acid and water. However, the whole process is not just due to the sulfuric acid; other key components are also at work within the battery. Industrially, sulfur dioxide is reacted with oxygen over a catalyst to make sulfur trioxide. This is cheaper but extremely difficult to do safely for the home chemist so the metabisulfite/oxidizer method is used instead. Aluminium, which is a highly reactive metal, will not dissolve in oxidizing acids (e.g. nitric), as it will grow on its surface a protective layer of aluminium oxide.
Filling your battery with water when the electrolyte level is low can maintain it healthy and safe to use. In the presence of air, sulphur dioxide is first obtained by burning the molten sulphur. In the presence of a catalyst for vanadium pentoxide, sulphur dioxide is then converted to sulphur trioxide.
With your calcium citrate separated, add just enough heavily diluted sulfuric acid to cover it and stir. Finish by filtering the solution with water into a beaker, heating it to evaporate any excess liquid, then pouring through a coffee filter to get your citric acid. In this video, i show as a first step how to make very clean diluted sulfuric acid by electrolysis of household products, then as a second step how to increa. Alternatively, you can buy a sulphuric acid solution with 1250 sp gravity from a battery shop to use as a battery electrolyte. How to make sulfuric acid by electrolysis of copper using an inert anode. Cut the onions into pieces to start the secretion process.
No comments:
Post a Comment